Acetate (CH3COO-) ion Lewis Structure, Resonance Structures
Acetate ion contains carbon, hydrogen and oxygen atoms. Lewis structure of CH3COO- contains a negative charge on one oxygen atom. There is a double bond in CH3COO- lewis structure. Two stable resonance structures also can be drawn for acetate ion.
CH3COO- ion lewis structure
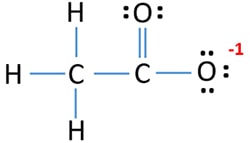
There is a double bond between carbon atom and one oxygen atom. Oxygen atom which has made a double bond with carbon atom has two lone pairs. Other oxygen atom has a -1 negative charge and three lone pairs.
Resonance Structures of CH3COO- ion
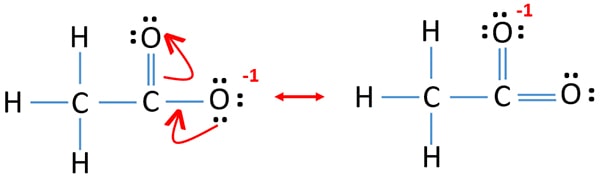
Two resonance structures can be drawn for acetate ion.
Steps of drawing lewis structure of CH3COO- ion
When we draw a lewis structure, few guidelines are given. Number of steps can be changed according the complexity of the molecule or ion. Because acetate ion is a simple molecule, it is extremely easy to draw the lewis structure. However those all steps are mentioned and explained in detail in this tutorial for your knowledge.
- Find total number of electrons of the valance shells of carbon, hydrogen and oxygen atoms
- Total electrons pairs existing as lone pairs and bonds
- Center atom selection
- Mark lone pairs on atoms
- Mark charges on atoms if there are charges.
- Check the stability and minimize charges on atoms by converting lone pairs to bonds to obtain best lewis structure.
Total number of electrons of the valance shells of acetate ion
There are three elements in acetate molecule; carbon, hydrogen and oxygen. Carbon is a group IVA element in the periodic table and contains four electrons in its last shell. Hydrogen, a group 1A element only has one electron and oxygen has six electrons in its last shell. Now, we can find out total number of electrons of the valance shells of acetate ion.
- valence electrons given by carbon atom = 4 * 2 = 8
- valence electrons given by hydrogen atoms = 1 * 3 = 3
- valence electrons given by oxygen atoms = 6 * 2 = 12
Because there is a -1 negative charge, an electron should be added to total number of electrons of the valance shells of acetate ion.
- Total valence electrons of acetate ion = 8 + 3 + 12 + 1 = 24
Total valence electrons pairs
Total valance electrons pairs = σ bonds + π bonds + lone pairs at valence shells
Total electron pairs are determined by dividing the number total valence electrons by two. For, acetate ion, total pairs of electrons are twelve in their valence shells.
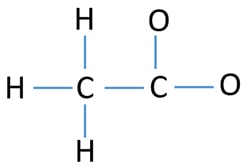
Center atom and skeletal of acetate ion
Skeletal of acetate ion is figured below. In a skeletal structure, atoms are only joint through single bonds and lone pairs are not marked.
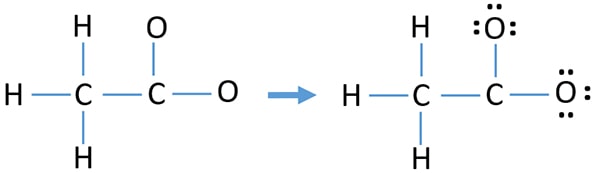
Mark lone pairs on atoms
After determining the skeletal of acetate ion, we can start to mark lone pairs on atoms. Remember that, there are total of twelve electron pairs.
- There is already six bond in the drawn skeletal. So, there are six remaining lone pairs to mark on atoms.
- Start to mark those remaining those lone pairs on outside atoms. As outside atoms, there are hydrogen and oxygen atoms. But, hydrogen atoms cannot keep more than two electrons in its last shell. Therefore, we cannot mark lone pairs on hydrogen atoms. Then, mark lone pairs on oxygen atoms. Each oxygen atom will take three lone pairs and all remaining six electron pairs are marked on both oxygen atoms.
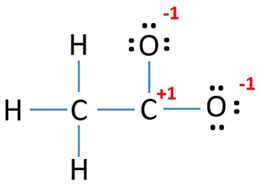
Mark charges on atoms
There are +1 charge on carbon atom and -1 charge on each oxygen atom.
Check the stability and minimize charges on atoms by converting lone pairs to bonds
Because, there are charges in above structure, we should try to reduce charges to get the most stable structure if possible.
It is possible to convert one lone pair of oxygen atom to make a bond with carbon atom as following.
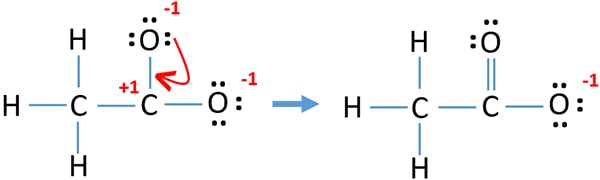
You can see now thee is only -1 charge on one oxygen atom. That means, this new structure is more stable than previous structure.
Questions