Formaldehyde (HCHO, CH2O, Methanal) Molecule Lewis Structure
Formaldehyde (HCHO) contains two hydrogen atoms, one carbon atom and one oxygen atom. There is a double bond between oxygen and carbon atom. Carbon atom is located as the center atom in HCHO.
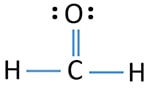
HCHO lewis structure
There are following specifications in the lewis structure of Cl2O5.
- Each hydrogen atom has made a single bond with center carbon atoms.
- Oxygen atom has made a double bond with carbon atom.
- When considering oxidation numbers, carbon atom is at 0 oxidation state.
Steps of drawing lewis structure of HCHO molecule
There are guidelines (several steps) given for drawing lewis structures. Number of steps can be changed according the complexity of the molecule or ion. There are important things to consider drawing the lewis structure of HCHO because there are seven elements in that molecule and it is bit complex molecule too. However, those all steps are mentioned and explained in detail in this tutorial to improve your knowledge.
- Find total number of electrons of the valance shells of carbon, hydrogen and oxygen atoms
- Determine total electrons pairs existing as lone pairs and bonds
- Center atom selection
- Mark lone pairs on atoms
- Mark charges on atoms if there are charges.
- Check the stability and minimize charges on atoms by converting lone pairs to bonds to obtain best lewis structure.
Total number of electrons of the valance shells of HCHO molecule
Hydrogen is a group IA element in the periodic table and contains only one electron in its last shell. Oxygen is a group VIA element in the periodic table and contains six electrons in its last shell. As well, carbon is IVA group element and has four electrons in its last shell. Now, we know how many electrons are there in valence shells of fluorine atoms.
- valence electrons given by hydrogen atoms = 1 * 2 = 2
- valence electrons given by oxygen atom = 6 * 1 = 6
- valence electrons given by carbon atom = 4 * 1 = 4
- Total valence electrons = 2 + 6 + 4 = 12
Total valence electrons pairs
Total valance electrons pairs = σ bonds + π bonds + lone pairs at valence shells
Total electron pairs are determined by dividing the number total valence electrons by two. For, HCHO molecule, Total pairs of electrons are six in their valence shells.
Center atom of HCHO molecule
Hydrogen atom cannot be a center atom because hydrogen atom cannot make more than 1 bonds. Because carbon's maximum valence (4) is higher than oxygen's maximum valence (2), carbon has the high chance to be the center atom. Also, carbon is more electropositive than oxygen, it is one more reason to select the carbon atom as the center atom.

Lone pairs on atoms
After determining the center atom and sketch of HCHO molecule, we can start to mark lone pairs on atoms. Remember that, there should be total of six electron pairs as lone pairs and bonds in valence shells.
- There are already three bonds in the above drawn sketch. So, there are three remaining lone pairs to mark on hydrogen, carbon and oxygen atoms.
- First, mark remaining lone pairs on outside atoms. We cannot mark lone pairs on hydrogen atoms because thee are two electrons already in hydrogen's valence shell. Therefore, remaining three lone pairs should be marked on oxygen atom. So, all remaining three electron pairs are marked and no more valence electron pairs to mark on carbon atom as lone pairs.
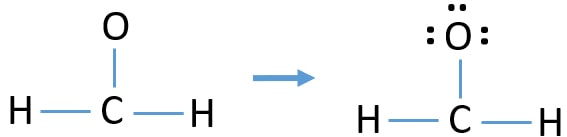
Mark charges on atoms
There are charges on oxygen atom and center carbon atom as following.
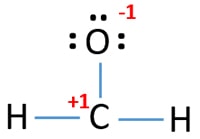
Check the stability and minimize charges on atoms by converting lone pairs to bonds
There are charges on center carbon atom and oxygen atom. We should check whether is there a possibility to reduce charges on atoms to obtain more stable structure (When atoms have less charges, stability of that molecule is high).
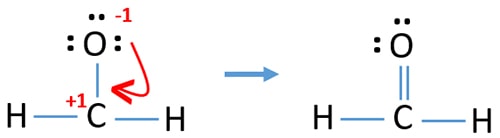
In the above structure, there are no charges on any atom. Therefore, we can think, we have obtained the lewis structure of HCHO.
Questions