Potassium permanganate and Hydrogen sulfide Reaction | KMnO4 + H2S
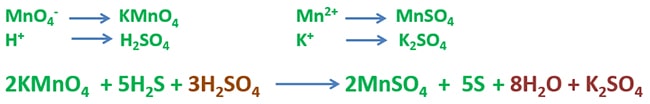
Potassium permanganate (KMnO4) reacts with hydrogen sulfide (H2S) and give manganese sulfate (MnSO4), sulfur, water and potassium sulfate. Manganese atom in KMnO4 is reduced to manganese +2 ion and sulfur atom in H2S is oxidized to sulfur. Purple colour of KMnO4 is reduced to colourless or pale pink colour during the reaction.
Written by: Heshan Nipuna, Eng., Chemical & Process Engineering, University of Peradeniya, last modified: 30/09/2021
Balanced reaction of potassium permanganate and sulfur dioxide reaction is given below.
2KMnO4(aq) + 5H2S(g) + 3H2SO4(aq) → 2MnSO4(aq) + 5S + 8H2O(l) + K2SO4(aq)
Manganese in potassium permanganate is reduced from +7 oxidation state to +2 oxidation state (Mn2+). As well, sulfur in hydrogen sulfide is oxidized from -2 oxidation state to 0 oxidation state (S).
Because oxidation numbers of several elements in reactants are changed when products are given, this reaction is considered as a redox reaction.
Reaction medium should be slightly acidified by adding few drops of sulfuric acid.
Physical observations during the reaction of potassium permanganate and sulfur dioxide
Dilute aqueous potassium permanganate solution is a purple colour solution. When colourless hydrogen sulfide gas is sent to that solution, purple colour of aqueous solution is decreased and become colourless or pale pink colour solution due to formation of Mn2+ cations. Also, you can see white - yellow colour precipitate (sulfur) is formed in the solution. Due to that sulfur particles, solution becomes unclear.
Safety during the reaction
Hydrogen sulfide is a highly toxic gas and acidic gas. Therefore, be careful to prevent any leakages during the treating hydrogen sulfide gas in to the potassium permanganate solution.
How to balance KMnO4 and SO2 reaction
Identify oxidation number difference of elements
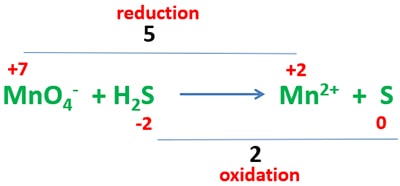
Because oxidation number of sulfur and manganese atoms are changed, identify what is the difference of oxidation numbers relevant to oxidation and reduction as below.
Exchange difference of oxidation numbers
Oxidation number difference of manganese atom is 5 and sulfur atom is 2. Then, exchange oxidation number difference as bellow.

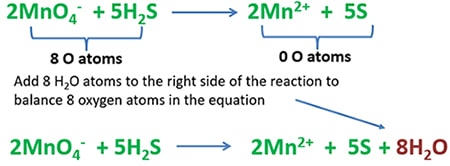
Balancing number of oxygen atoms
Count the total number of oxygen atoms in the left side and right side separately. To balance the number oxygen atoms, add 8 H2O molecules to the right side od the reaction.
Balancing number of hydrogen atoms
Count the total number of hydrogen atoms in the left side and right side separately. To balance number hydrogen atoms, add 4 H+ ions to the right side.
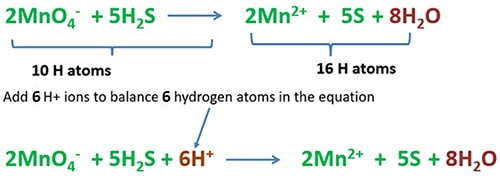
Now, we have obtained the balanced ionic equation of KMnO4 and H2S reaction.
Balancing chemical equation
Now, we can balance the ionic equation and find chemically balanced equation.